How AI Can Transform Modern Pharma QC Environments

Commissioning, qualification, and validation (CQV) sit at the heart of quality and regulatory compliance within the life sciences. Yet, for many QC teams, the process still feels stuck in a paper-first past. Protocols are written in Word, emailed, printed, signed, scanned, and then filed. However, errors are often discovered too late, and documentation becomes disconnected from real-world lab work. The result is slower product release, increased risk, and mounting pressure on already stretched QC and validation groups in large pharma organizations.
At the same time, QC environments are now more complex than ever. Teams must qualify and maintain an ever-expanding ecosystem of instruments, software, and digital systems all whilst under the scrutiny of 21 CFR Part 11, EU GMP Annex 11, and data integrity expectations such as ALCOA. Doing that with manual, paper-based CQV makes it harder to prove control, ensure complete audit trails, and maintain confidence in every signature, timestamp, and protocol version.
That is now changing. Driven by shorter timelines, stricter regulatory expectations, and the push toward fully digital labs, CQV is undergoing a well-needed fundamental shift – from paper to structured data, and from static documentation to intelligent, AI-enabled systems validation.
A three-phase journey to intelligent CQV
- Phase 1: Digitalization of CQV workflows
Paper records, folders, and unstructured files are replaced with validated, digital CQV platforms. QC teams gain consistent templates, secure audit trails, controlled access, version management, and real-time visibility of protocol execution all within a compliant framework designed to meet 21 CFR Part 11 and EU Annex 11 expectations. - Phase 2: Process automation and standardization
Once CQV activities are digital, logic and rules can begin to automate routine process steps. Approvals, test execution, trace-matrix creation, status tracking, and error checks can be configured into the system, reducing manual rework and enforcing standard practices across sites, products, and instruments. - Phase 3: AI-augmented, intelligent validation
With structured, high-integrity historic data in place, AI and machine learning can be safely introduced. Trained models help detect anomalies, guide real-time execution decisions, and provide predictive insights that support risk-based CQV planning and continuous assurance.
Most organizations today are still in phase one or two (supported by TotalLab’s AuditSafe software in some cases), building the digital and procedural foundation that AI depends on. But those that complete the transition to structured, validated CQV data are positioned to unlock the next level of intelligent, data-driven validation in partnership with TotalLab.
Recent regulatory guidance shows that the shift to the third phase aligns with changing oversight expectations – it’s what the regulators want to start seeing in large pharmaceutical companies. The US Food and Drug Administration (FDA) has already introduced draft guidance supporting the use of AI or machine learning in drug development, emphasizing risk-based validation and transparency. Matching that, documents from the European Medicines Agency (EMA) and Pharmaceutical Inspection Convention Co-operation Scheme (PIC/S) encourage digital maturity and proactive quality control. The message from the regulators is clear – AI and ML are welcome as long as they are implemented with sufficient control mechanisms in place.
Why AI-Enabled CQV Matters for Pharma QC
A digitally transformed CQV function does far more than just “go paperless.” For QC teams in large pharma, it creates a controlled, compliant, and traceable environment in which validation becomes a valuable source of future process insight rather than just a regulatory obligation.
With digital CQV platforms in place, QC leaders benefit from:
- Real-time status tracking of commissioning, qualification, and validation across multiple systems and sites.
- Consistent use of approved protocols and templates, reducing variability and the risk of missing steps or signatures.
- Built-in audit trails capturing who did what, when, and why, supporting inspections and internal QA reviews.
- Direct alignment with ALCOA principles, ensuring data are attributable, legible, contemporaneous, original, and accurate – even in challenging environments.
Most importantly, documentation stops being a backlog of static binders and becomes an asset that QC teams can search, visualize, and act on in real-time.


Bringing AI into CQV: From documents to decisions
Once digital, validated CQV data are available at scale, AI can begin to add real value for QC teams. In practice, AI is typically already supporting CQV in three main domains: smart documentation, real-time execution support, and predictive quality.
1. Smart documentation and faster validation reporting
Generative AI models trained on validated templates can now support drafting of protocols, test scripts, and reports. These tools use language generation to populate fields, apply standard formatting, and incorporate references to historical data, which helps enforce consistency in terminology and structure across all validation documents.
In real deployments, AI validation assistants have reduced document preparation time from weeks to minutes, delivering up to faster document creation and significant cost savings. AI tools can automatically attach supporting evidence to projects, such as instrument printouts and screenshots as they are uploaded, while also applying validation rules and checklists in real-time to flag omissions or errors.
Using computer vision (another area of expertise for TotalLab) and natural language processing, these systems can cross-check screenshots and instrument outputs against expected results, highlighting mismatches or missing approvals for review. For QC teams running large numbers of similar qualifications, this reduces human review effort and helps maintain accuracy at high volumes.


2. Execution with real-time, data-driven assistance
Validation execution is evolving from a static checklist to a dynamic, data-guided process. With IoT sensor streams and AI models running in the background, systems can detect deviations as they occur, rather than days later during document review.
For example:
- Pressure changes during equipment qualification can be flagged instantly for investigation.
- Borderline test results can trigger recommended retesting or additional checks.
- Repeated deviations across sites or equipment types can be surfaced as patterns automatically for QC leaders to address.
Machine learning also enables adaptive execution. Based on real-time test outcomes and preconfigured decision logic, an AI system may recommend repeating specific steps or skipping unnecessary ones whilst remaining always within predefined, risk-based boundaries approved by human QA teams. This reduces downtime, removes guesswork, and supports consistent decision-making in time-sensitive QC environments.

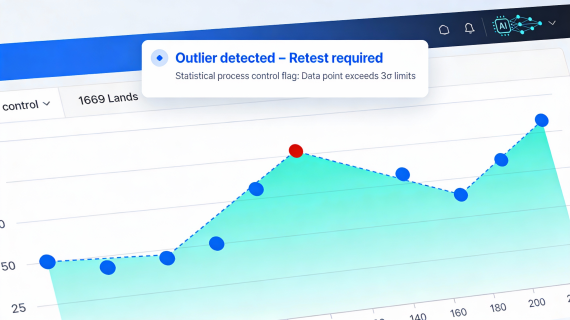
3. Predictive quality and risk-based CQV planning
Perhaps the most transformative use of AI in CQV is predictive quality management. AI models can ingest historical qualification data, execution logs, and sensor readings to detect subtle patterns that precede deviations – the early warning signs that busy QC teams rarely have time to analyze manually.
A gradual drift in vibration data, for example, may foreshadow a calibration failure in six weeks. By flagging that drift early, AI allows QC teams to schedule preventive maintenance or refine test protocols before nonconformances occur. TotalLab already has non-AI enhanced software to spot trends such as this – TrendLab – manually, however, an AI-empowered model could be custom-generated for your needs and your site.
Over time, this approach shifts validation from one-off, project-based activities toward continuous assurance. Qualified equipment and validated systems remain under AI-enabled surveillance for their entire life cycle, with custom models learning from every executed protocol, deviation, and result to improve recommendations.

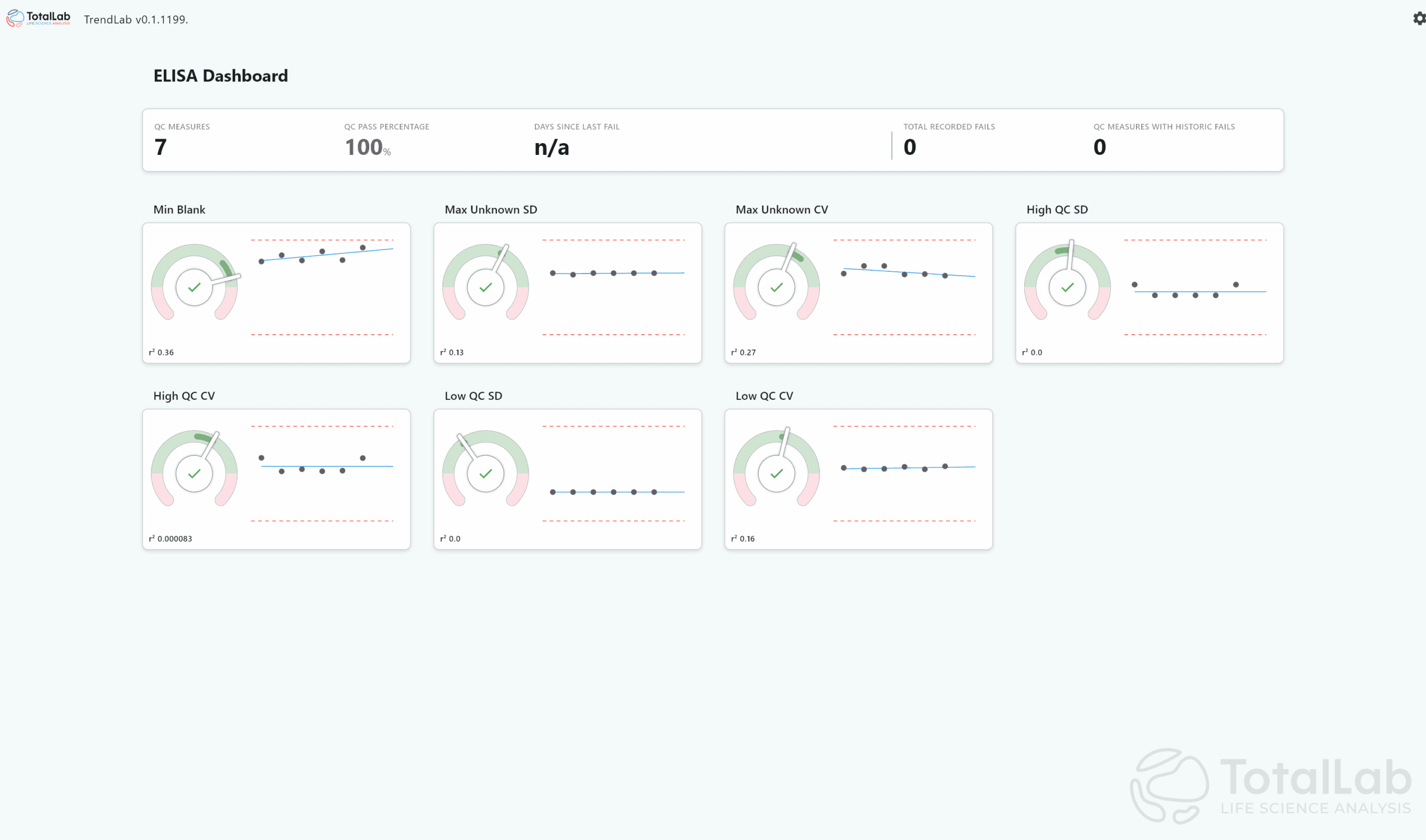
Building Trustworthy AI for CQV Under Regulatory Scrutiny
For QC and QA leaders in large pharma, one question is central: how can AI be used in CQV without compromising compliance? Regulators are increasingly explicit that AI is welcome (see the reference links above) – provided it is implemented with control, transparency, and robust validation. By partnering with TotalLab and leveraging our decades of experience in 21 CFR Part 11/GMP-compliance, QC and QA leaders can drastically de-risk such a project.
Current guidance emphasizes a risk-based approach to AI use in regulated environments, including:
- Tracking model versions and training data to ensure traceability.
- Verifying model performance (e.g., false-positive and false-negative rates) and documenting limitations.
- Recording all AI-generated alerts, recommendations, and human overrides.
- Embedding AI outcomes into audit-ready records that can be reviewed during inspections.
Frameworks such as the updated GAMP 5 Guide and PIC/S guidance on data management and computerized systems provide practical direction on AI governance and validation. AI does not replace CQV protocols; it strengthens them by increasing visibility, consistency, and predictive capability, as long as the underlying systems are designed, qualified, and monitored appropriately using something like the custom overview dashboard shown on our AI Software Development page:

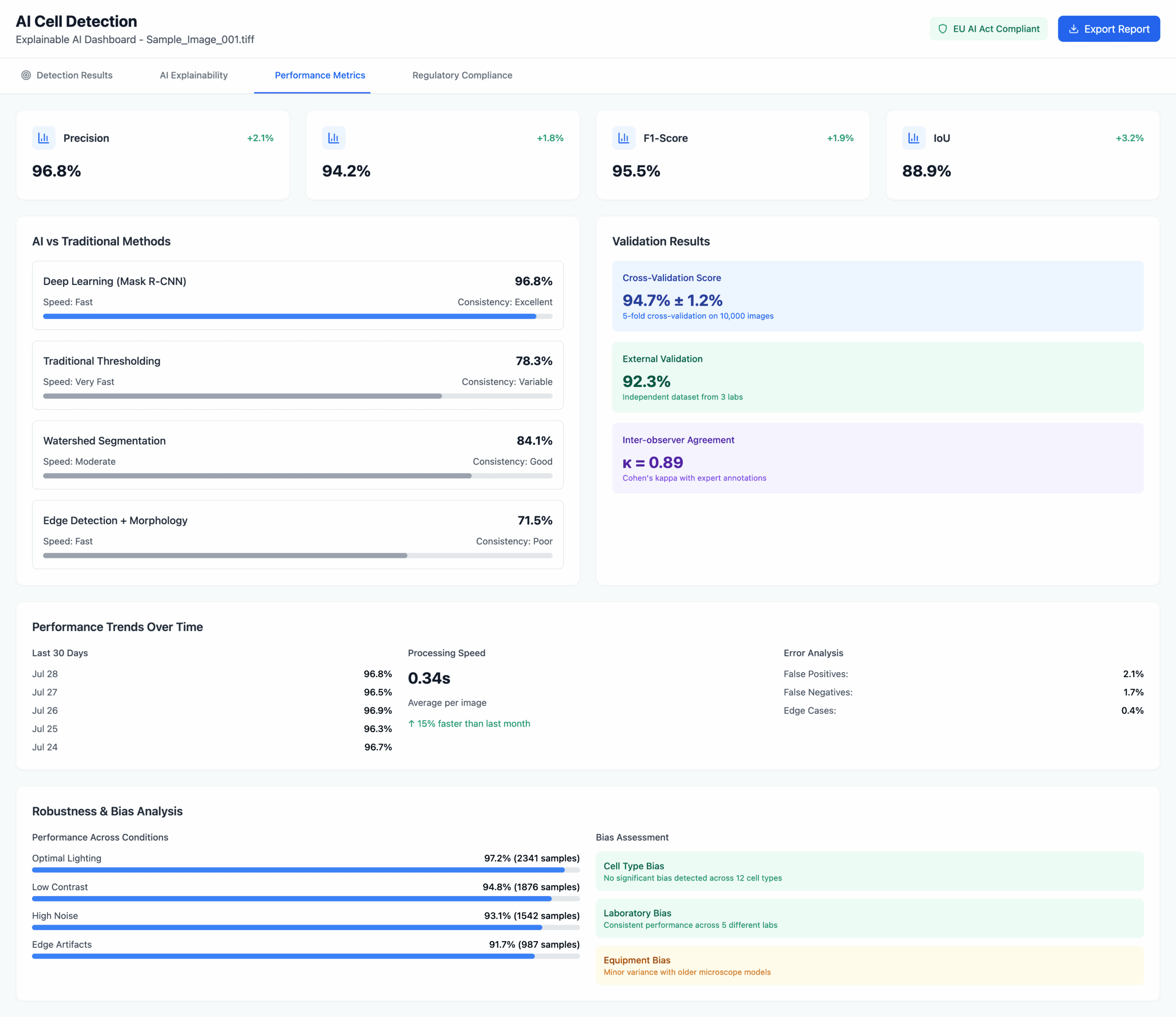
What comes next for CQV in QC labs?
As digital maturity increases across QC and manufacturing, AI is poised to act as a continuous layer of quality oversight in CQV. Validation will move from isolated, document-heavy projects toward integrated, intelligence-driven operations that support faster, more confident decision-making.
For QC teams, AI-enabled CQV will help to:
- Prevent deviations before they occur through early signal detection and pattern recognition.
- Prioritize validation scope based on historical risk and equipment performance, focusing effort where it matters most.
- Auto-generate documentation and fill evidence gaps during execution, reducing administrative burden.
- Perform continuous, automated audit checks that reduce the volume of manual review.
In that model, AI does not remove control from QC; it enhances it. Quality teams retain authority over decisions while AI surfaces insights and automates repeatable tasks, turning routine validation into a source of actionable intelligence.
Once organisations have performed the groundwork by going digital and standardizing workflows, the next step is similar: consolidating CQV into structured, validated systems that prepare the organization for intelligent validation. From there, activating AI becomes an evolution of an existing practice – not a leap into the unknown.
If you’re ready to start your AI CQV journey today, why not have a chat with us to see what TotalLab can do for you!

