GMP & 21 CFR Part 11 Compliant Gel Analysis Software | Phoretix 1D

Ensure Compliance, Accuracy, and Efficiency in Every 1D Gel Analysis
Our Phoretix 1D software is already the complete solution for analyzing 1D SDS-PAGE gel and immunoblot images, but when also combined with our AuditSafe software, it allows users to meet and even exceed FDA 21 CFR part 11, EudraLex Annex 11, GAMP4 or more general GMP standards/ALCOA principles for compliance within regulated or QC/QA environments, from captured image all the way through to final analysis and report production.
Phoretix 1D + AuditSafe is the trusted solution for regulated laboratories that demand 21 CFR Part 11 and GMP compliance in protein gel, SDS-PAGE, and Western blot analysis. Achieve audit-ready results, secure data integrity, and effortless regulatory reporting—all in a single, automated platform.

Our Phoretix 1D software is already trusted by the world’s leading educational, industrial and pharmaceutical companies:

Why Choose Phoretix 1D for Regulated Labs?
Combine all the benefits of our Phoretix 1D gel and immunoblot image analysis software:
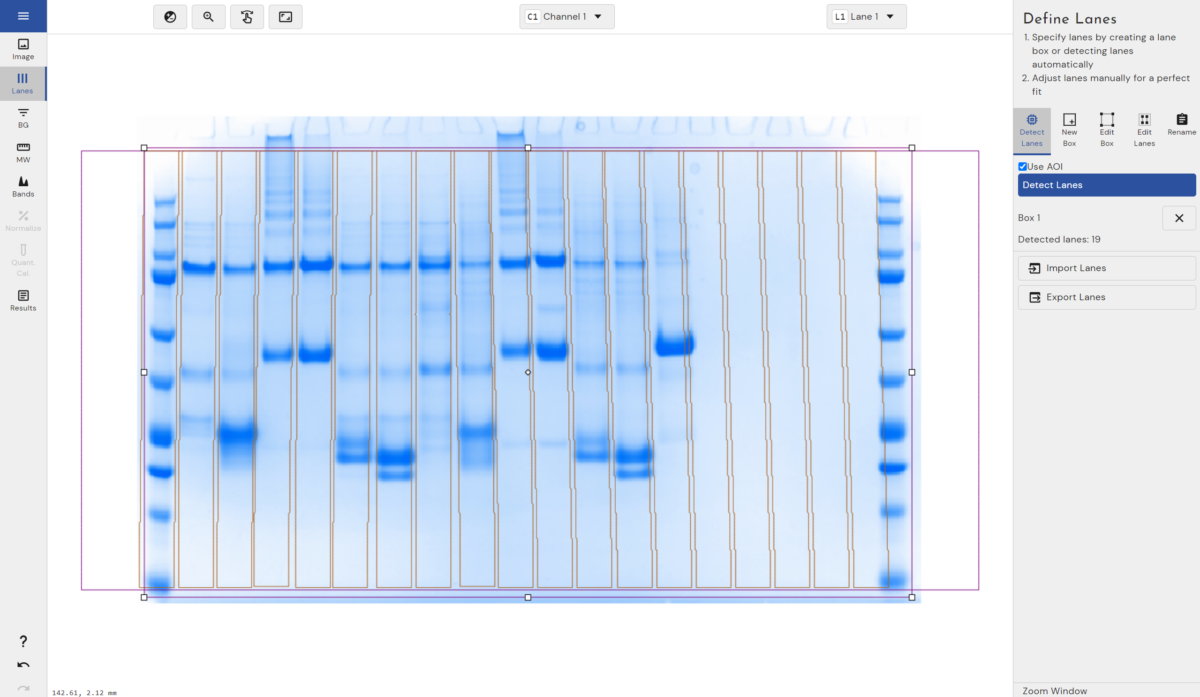
- Automated Lane and Band detection
- Background subtraction to remove noise and increase accuracy
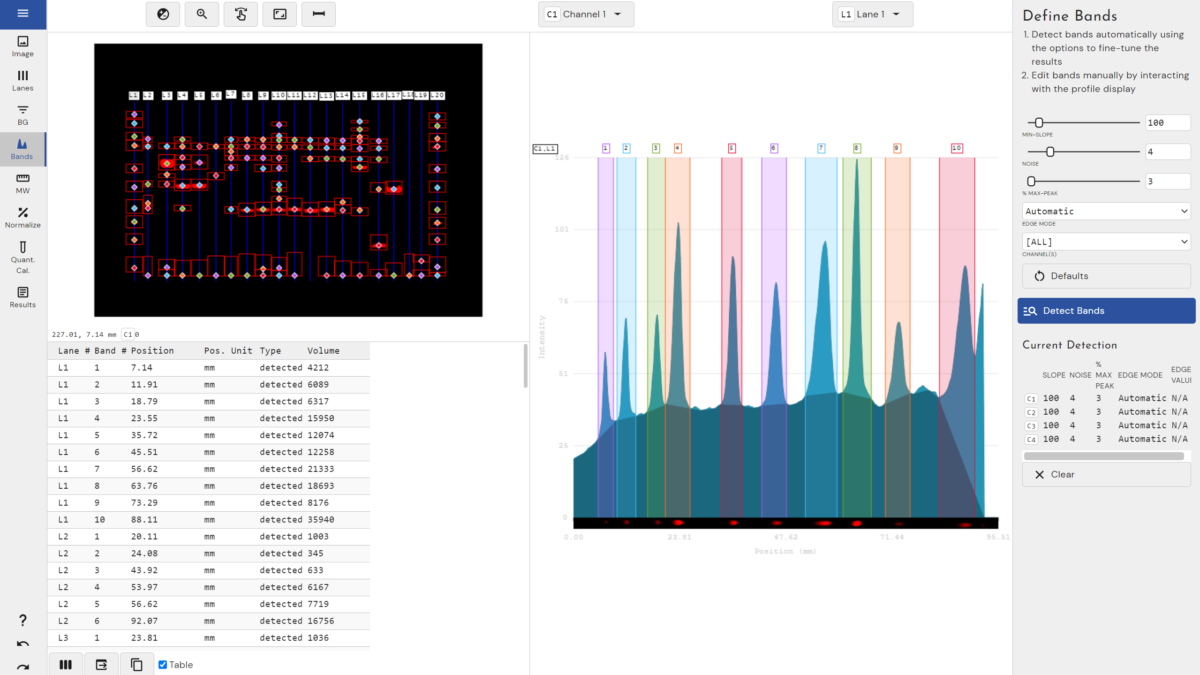
- Automatic band detection
- Molecular Size calculation
- Quantity Calibration
- Normalization of bands to loading controls
With our AuditSafe compliance module, which provides the following additional features:
- Robust password control which can be configured to follow local password policy or integrate with networked Windows logins/Active directory (21 CFR 11, 11.200 (a))
- Audit trails with complete user activity, login attempts, and detailed analysis logs (21 CFR 11.10(e))
- User groups with granular access levels based on role and responsibilities
- Limited system access (21 CFR 11.10(d))
- Electronic signatures for approvers with check-in and check-out process for data archiving and approval (21 CFR 11, Subpart B)
- Emergency logins (21 CFR 11, 11.200 (3))
- Version History (21 CFR 11, 11.10 (k))
- Image calibration
- Easy integration with existing enterprise level data backup solutions
Already Trusted by Leading Pharma & Biotech Labs
- Proven in GMP and GLP environments worldwide for over 12 years
- Our software reduces operator error and increases reproducibility in QC workflows