How SpotMap MS Identifies High-Risk Host Cell Proteins (HCPs) in Biopharmaceutical Analysis
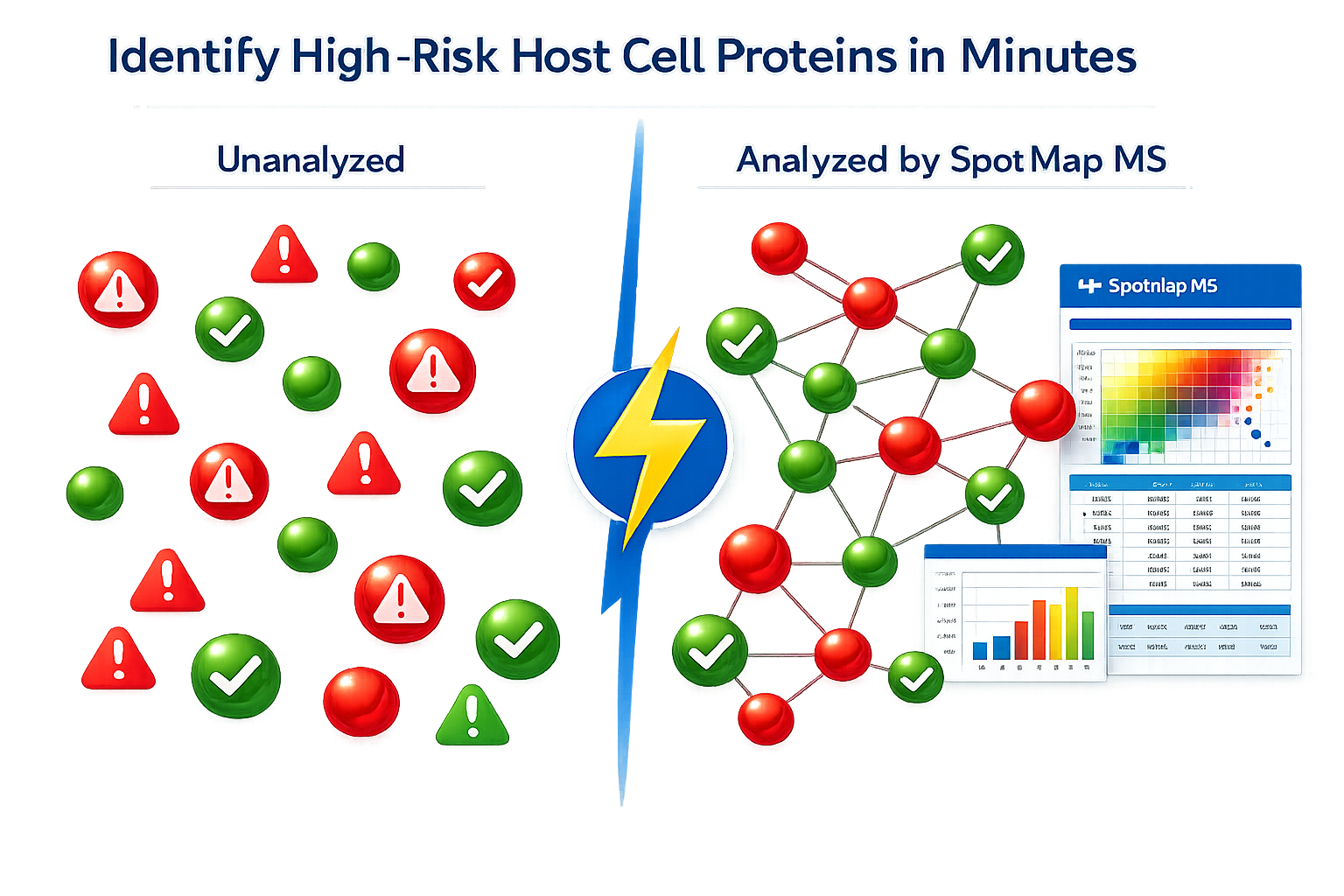
Understanding Automated HCP Risk Assessment in LC-MS Workflows
SpotMap MS uses advanced automated analysis to identify dangerous host cell proteins directly from your LC-MS data. Unlike traditional manual approaches, SpotMap MS leverages comprehensive HCP databases and intelligent protein matching algorithms to flag high-risk HCPs automatically—reducing analysis time and improving accuracy.
Key Features & Differentiators
1. Intelligent AI-Powered HCP Database Technology
SpotMap MS includes an inbuilt, free database, created by TotalLab, of known high-risk host cell proteins, which was generated using the latest AI models by scouring the latest biopharmaceutical research and collating that information. Unlike the very popular BioPhorum Free HCP Database, which has not been updated since 2021, the TotalLab HCP database contains nearly twice as many HCP entries and was last updated in Q4 2025.
SpotMap MS automatically compares detected peptide sequences from your LC-MS data against our comprehensive database to identify potentially dangerous proteins – eliminating the need for manual cross-referencing or users having to provide their own database. However, SpotMap MS also supports users’ existing host cell protein database if they do have one already. Please contact us if you want to explore importing your existing database/HCP reference data into the software.
2. Automated High-Risk Protein Detection
Unlike competitors, SpotMap MS automates the identification of potentially problematic HCPs. The software highlights proteins that pose immunogenicity risks, drug product stability threats, and regulatory compliance issues – all without manual intervention.
3. Reduced Manual Workflow Burden
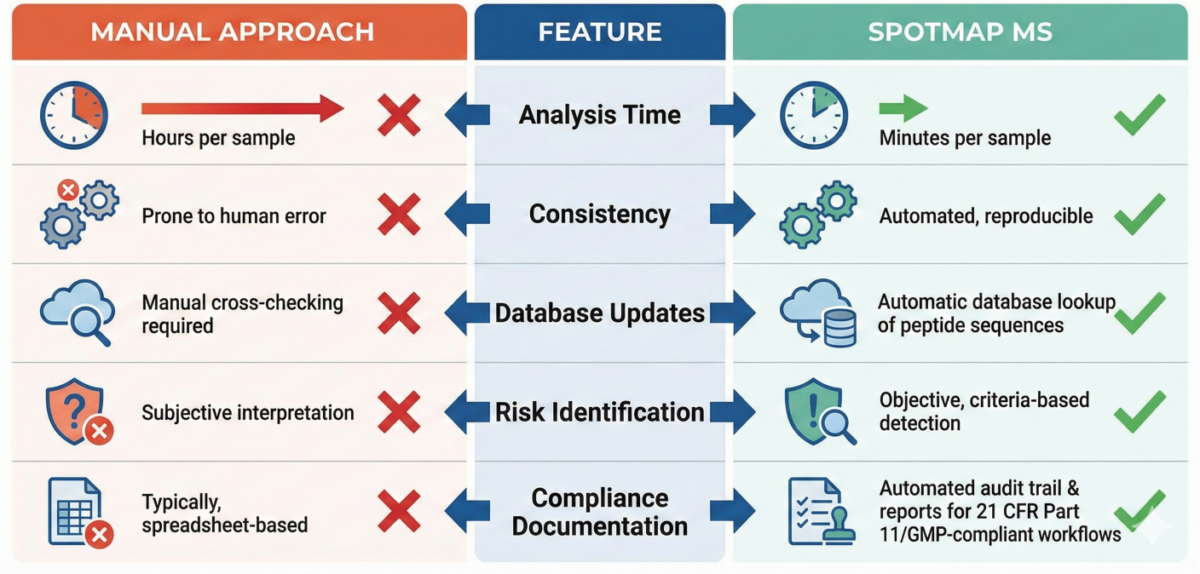
Traditional methods require scientists to manually detect HCP proteins in their data, then cross-match them against reference databases – a time-consuming, error-prone process. SpotMap MS automates this entire workflow, enabling labs to identify high-risk HCPs with minimal human intervention, improving reproducibility and throughput for time-sensitive projects.
How SpotMap MS Works
Step 1: LC-MS Data Analysis
Import your liquid chromatography-mass spectrometry (LC-MS) results into SpotMap MS. The software processes protein identifications and peptide sequence data from your experiment.
Step 2: Automatic HCP Matching
SpotMap MS compares all detected proteins and peptides against its inbuilt high-risk HCP database (or your own if imported). The algorithm identifies exact and potential matches in seconds.
Step 3: Risk Assessment
Detected HCPs are classified by risk level – from potentially problematic to high-risk – based on their immunogenicity potential, stability impact, and regulatory relevance. The software flags entries requiring further investigation.
Step 4: Reproducible Reporting
Generate publication-ready reports documenting which HCPs were detected, their risk classification, and their abundance in your sample. Perfect for regulatory submissions and clinical documentation.

Why SpotMap MS Outperforms Manual Analysis
Use Cases & Applications
- Biopharmaceutical QC Testing: Rapidly screen therapeutic proteins for dangerous HCP presence
- Process Development: Monitor HCP levels across purification method variations
- Regulatory Compliance: Generate evidence for FDA submissions proving HCP control
- CMC Documentation: Build comprehensive HCP analysis packages for drug approvals
- Research & Academia: Quantify host cell protein expression in recombinant protein production
Key Benefits for Your Lab
✓ Save Hours Per Sample – Automate HCP detection across dozens of samples simultaneously
✓ Eliminate Manual Errors – Objective, reproducible results independent of operator skill
✓ Meet Regulatory Standards – Generate compliant documentation for FDA 21 CFR Part 11 submissions
✓ Reduce Outsourcing Costs – Bring HCP analysis in-house without additional staff training or costly outsourcing
✓ Stay Current – Access a newer, updated HCP risk databases reflecting latest research
Frequently Asked Questions
What databases does SpotMap MS support?
SpotMap MS includes an AI powered, TotalLab developed, host cell protein database by default free of charge with nearly double the number of entries of the BioPhorum free HCP database. Users can also integrate custom HCP reference databases or select alternative databases based on your specific analytical needs.
Can SpotMap MS work with my existing LC-MS data?
Yes. SpotMap MS accepts data from standard LC-MS workflows and is compatible with common protein identification formats. No instrument modifications required.
How does SpotMap MS compare to manual database searches?
Manual approaches require scientists to manually identify proteins in LC-MS data, then cross-reference them against HCP databases. This is time-consuming, subjective, and prone to errors. SpotMap MS automates this entire process in minutes.
Is SpotMap MS suitable for regulatory submissions?
Absolutely. SpotMap MS can be integrated into TotalLab’s existing AuditSafe 21 CFR Part 11/GMP-compliance software to generate audit trails, reproducible results, and compliant documentation ideal for FDA submissions and clinical dossiers.
Get Started with SpotMap MS
Try SpotMap MS Free Today
Experience how SpotMap MS transforms your HCP analysis workflow. Our team provides full technical support and personalized demos for pharmaceutical QC, biotech R&D and regulatory teams.
