Add 21 CFR Part 11 Compliance to Your Instruments in Weeks, Not Years | AuditSafe

A plug-and-play compliance middleware that instantly adds electronic signatures, secure audit trails, and Active Directory integration to your commercial lab equipment.
Unlock Regulated Pharma Markets Without the R&D Nightmare
If you manufacture laboratory hardware or software, you already know that selling into GMP-regulated environments requires strict adherence to FDA 21 CFR Part 11 and EU Annex 11.
But building these compliance features from scratch is a massive drain on your resources. It forces your engineering team to spend 18-24 months building security layers, user access controls, and audit trails instead of innovating on your core technology.
AuditSafe is your shortcut. By integrating our proven compliance middleware, you can instantly upgrade your product line, qualify for enterprise pharma bids, and get to market faster.
TotalLab’s AuditSafe software is already trusted to secure hardware and software from the following OEM Partners:

Why Partner with TotalLab for OEM Compliance?
- Accelerated Time-to-Market: Bypass the years of software development, testing, and regulatory validation. AuditSafe drops into your existing architecture seamlessly.
- Expand Your Total Addressable Market (TAM): Stop getting disqualified by QA Directors. Instantly open your sales pipeline to highly regulated biotech, pharma, and clinical manufacturing labs.
- Flexible Licensing Models: Whether you want to bundle AuditSafe with every instrument, offer it as an optional premium upgrade, or explore a fully white-labeled solution, we structure our licensing to fit your exact business model.
- Vendor-Neutral Architecture: AuditSafe is designed to wrap around almost any PC-based laboratory system, meaning you don’t have to rewrite your underlying proprietary software.
| Strategic Factor | Building Compliance In-House (The Old Way) | Using AuditSafe (The Smart Way) |
| Time to Market | 18 – 24 Months. Severe delays to your product launch roadmap. | Weeks. Plug-and-play middleware gets you selling immediately. |
| Development Cost | High & Unpredictable. Requires a dedicated team of specialized security developers. | Predictable ROI. Flexible licensing models scaled to your sales volume. |
| Regulatory Validation | High Risk. You have to build, test, and validate complex ALCOA+ features from scratch. | Zero Risk. A proven, industry-tested solution already trusted by regulated labs globally. |
| Ongoing Maintenance | Resource Drain. Your engineers are stuck updating compliance patches forever instead of innovating. | Managed for You. TotalLab handles all software updates and shifting regulatory requirements. |
| Core Business Focus | Distracted. Your team is focused on IT security and Active Directory integration. | Focused. Your team focuses 100% on what you do best: scientific hardware and analysis. |
Out-of-the-Box ALCOA+ Capabilities
When you integrate AuditSafe, your instruments instantly inherit:
- Centralized Security: Active Directory / LDAP integration for seamless, IT-approved user management.
- Data Locking: Protection against the deletion or unauthorized modification of your instrument’s raw data files.
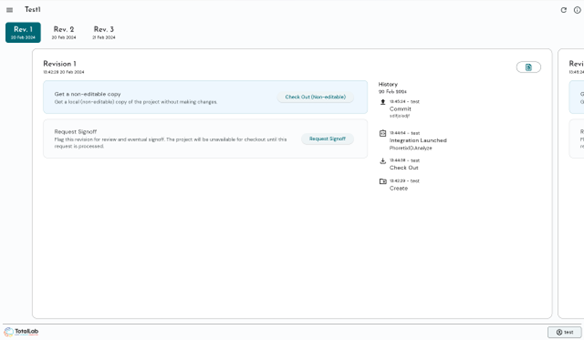
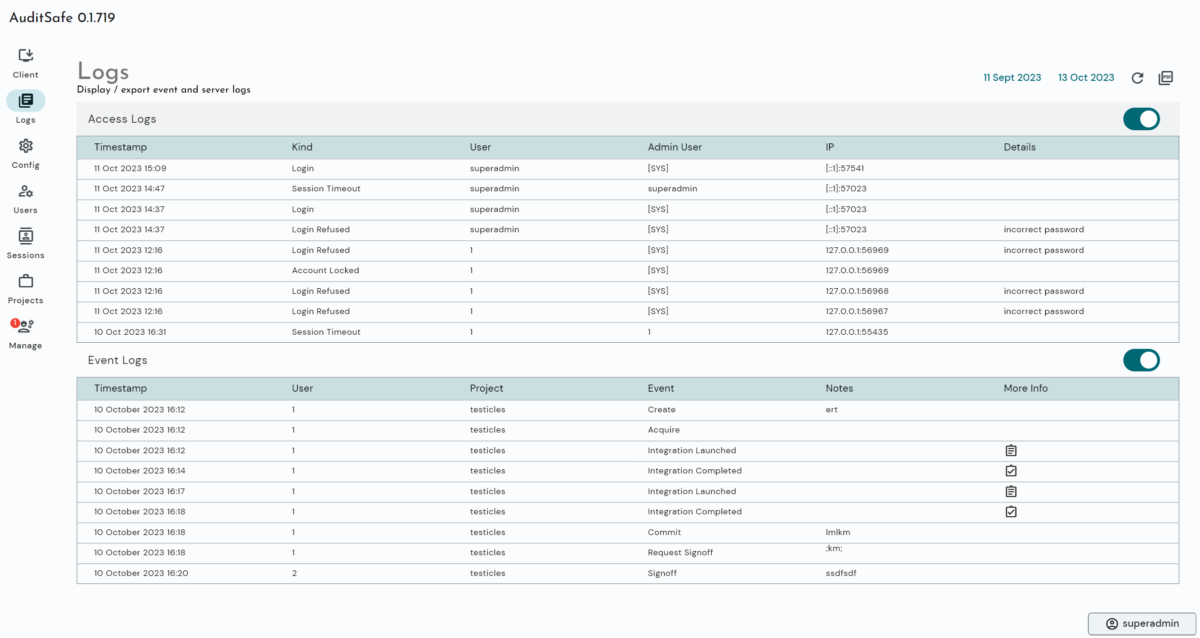
- Automated Audit Trails: Invisible, time-stamped background logging of every user action and parameter change.
- Electronic Signatures: Compliant sign-off workflows embedded directly into the digital process.
Ready to Upgrade Your Product Line?
Let’s talk integration. Fill out the form below, and our OEM Partnerships Director will be in touch within 1 business day to schedule a technical discovery call. We’ll discuss your specific instrument architecture and map out the fastest path to compliance.