LC-MS HCP Software Comparison: SpotMap MS vs Proteomics-Based Tools
Most LC-MS software your team is using right now wasn’t designed for host cell protein analysis. It was designed for proteomics, and pressed into service for HCP. This LC-MS HCP software comparison looks at where that legacy shows up — and what changes when the software is built for HCP from the ground up.
Why LC-MS HCP analysis is still stuck in the lab
The technique is mature. The instruments are extraordinary. The bottleneck is the software. Existing LC-MS analysis platforms were designed for large-scale proteomics — and that legacy is the single biggest reason routine, GMP-environment LC-MS for HCP hasn’t taken off the way the technology should allow.
Five problems show up consistently when analytical development and QC teams describe their current workflow:
1. Software designed for proteomics, not HCP
Current LC-MS analysis software is built for proteome-scale research questions. HCP analysis is a different problem — and treating it as a proteomics edge-case shows in the workflow, the output, and the validation burden that lands on the analyst.
2. Too many parameters
To handle every research question proteomics throws at it, generic software exposes long lists of parameters. That flexibility comes at a cost: different analysts running the same data routinely produce different results.
3. Validation eats the analyst
Proteomics-based tools tend to output a list of identified proteins and relative abundances. Validating those matches against your HCP context — manually, spectrum by spectrum — can take as long again as the analysis itself.
4. Manual, laborious data review
LC-MS as a technique is mature. The data analysis layer hasn’t caught up. Despite the maturity of the instrument science, day-to-day HCP analysis is still time-intensive, manual, and easy to get wrong.
5. Few GMP-compliant options
The combined effect of the above is that LC-MS for HCP rarely makes it from R&D into the regulated QC environment where it’s most needed. Very few platforms support 21 CFR Part 11 / EudraLex Annex 11 routinely. Customer choice has been narrow – until now.

SpotMap MS vs proteomics-based alternatives: a side-by-side LC-MS HCP software comparison
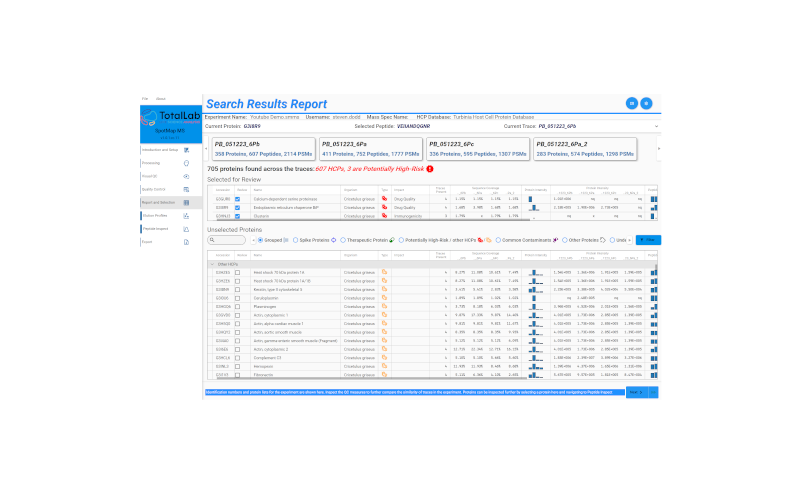
SpotMap MS was built ground-up for host cell protein analysis. The table below compares it against the three most common proteomics-based platforms used for HCP analysis today — Protein Metrics’ Byos and Byonic, Thermo Fisher’s BioPharma Finder, and Genedata Expressionist alongside academic tools like MaxQuant. The comparison is drawn from each vendor’s publicly documented capabilities at time of writing.
| Capability | SpotMap MS (TotalLab) | Byos / Byonic (Protein Metrics) | BioPharma Finder (Thermo Fisher) | Genedata Expressionist / MaxQuant |
|---|---|---|---|---|
| Designed for | HCP analysis, ground-up | General proteomics & biopharma characterisation | Biotherapeutic characterisation, broad MS workflows | Discovery-scale proteomics, omics |
| Setup to first result | Point at FASTA + raw files. Done. | Workflow building required | Method-specific configuration | Heavy parameter setup |
| HCP threat-level scoring | Built in. Pre-loaded HCP risk database. | Not a native feature | Not a native feature | Not a native feature |
| Pre-loaded HCP libraries (CHO, E. coli, HEK) | Yes - plus user / proprietary FASTA | User-built libraries | User-built libraries | User-built libraries |
| Visual QC of replicates | Native ion intensity maps | Workflow-dependent | Workflow-dependent | Workflow-dependent |
| Reproducibility across users | QC-first - minimal parameters | Strong, but parameter-heavy | Method-template dependent | Highly parameter-sensitive |
| 21 CFR Part 11 / Annex 11 | Via AuditSafe wrapper. Full audit trail, e-sigs. | Compliance-ready editions available | Compliance-ready editions available | Limited / not native (especially MaxQuant) |
| USP alignment | Designed against the chapter | Possible, configuration-dependent | Possible, configuration-dependent | Possible, configuration-dependent |
| In-person IQ/OQ available | Yes - included with regulated versions | Vendor-dependent | Yes, fee-based | Generally not |
| Built with HCP community | Long-time BEBPA HCP Conference supporter | Broader proteomics community | Broader biopharma community | Academic proteomics roots |
Vendor and product names are trademarks of their respective owners. The comparison reflects publicly documented capabilities and may not reflect customisations available through individual vendor agreements.
What “built for HCP” actually buys you
Three concrete things change when LC-MS HCP analysis software starts from HCP rather than ending up at it. Each one is a workflow your QC team is doing today, manually, by hand, in spreadsheets and side documents.
Set it, then forget it
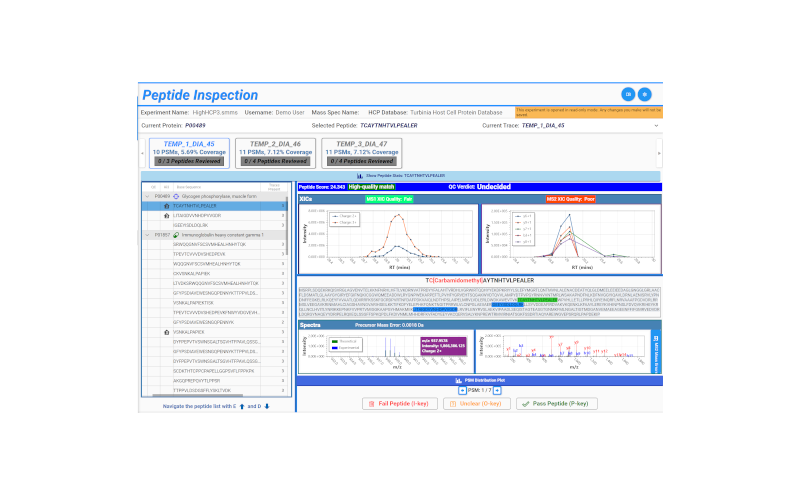
Specify your FASTA file and your raw MS trace files. SpotMap MS performs a fully automated HCP analysis end-to-end — no method-building, no hand-holding, no babysitting. Walk away. Come back to a comprehensive HCP report based on data dependent acquisition (DDA) methodology, ready for review.
AI-powered threat scoring
The TotalLab HCP database scores identified host cell proteins by published threat level — drawn from peer-reviewed literature on HCPs known to be problematic in biotherapeutics. Your output stops being “here are 200 proteins” and starts being “here is the one you need to look at first.” Pre-loaded libraries are available for the most common host cell lines including CHO, E. coli and HEK.
Visual QC that catches what you’d miss
Ion intensity maps surface failed technical replicates and acquisition issues at a glance — before you spend hours analysing data that was never going to be valid. Issues that other software has no way of showing become obvious in seconds.
What HCP analysts say about SpotMap MS
SpotMap MS is in production at some of the world’s largest biotherapeutic developers. Two of the things customers tell us most often:
“Typically with proteomics-based software to search against the CHO proteome, we don’t have much functionality to validate spectra. As a result, the output becomes ‘here is a list of proteins we identified and their relative abundance’ — if we wanted to validate those matches it could easily take twice as long without SpotMap MS.”
— Senior pharmaceutical analytical scientist“Without SpotMap MS and its Visual QC functionality we didn’t even realise one of our technical replicates had failed — our current software had no way of showing it.”
— Biopharmaceutical QC team
Audit-ready compliance, in every regulated jurisdiction
When SpotMap MS is paired with TotalLab’s AuditSafe wrapper, you get a fully audit-trailed, e-signed, GxP-ready environment for routine HCP analysis. The same wrapper is already in production at major pharma companies for their wider regulated software stack.
AuditSafe restricts software and data access through secure user sign-ins, granular permissions, audit logs, electronic signatures and image authenticity checks. It is designed to meet:
– FDA 21 CFR Part 11
– USP <1132.1> (Residual Host Cell Protein Measurement in Biopharmaceuticals)
– EudraLex Annex 11
– ISPE GAMP and ALCOA+ data integrity principles
For regulated installations, TotalLab provides an in-person IQ/OQ package as part of the software validation pathway.
Who SpotMap MS is built for
SpotMap MS is designed for four kinds of teams in particular.
Pharma QC and Analytical Development
Running routine HCP screening on biotherapeutic batches. Need reproducibility across analysts, sites and shifts. Need an audit trail. Cannot afford a different result depending on who ran the analysis.
Bioprocess Development
Screening clones, optimizing process steps, characterizing new biotherapeutics. Need to identify and quantify HCPs fast, and need to flag the high-risk ones before they become a CMC problem downstream.
CDMOs and service labs
Running HCP campaigns for multiple sponsors, often against tight turnaround. Need a tool that can move from method to result without the analyst owning every parameter, and that delivers a regulator-ready report on the way out.
Teams adding LC-MS to ELISA
Already running HCP ELISA. Need orthogonal LC-MS coverage to characterize antibody reactivity and pick up HCPs the assay can’t see. SpotMap MS pairs with SpotMap 2D and PlateLogic for the full HCP picture.

Frequently asked questions about LC-MS HCP software
Does SpotMap MS work with our existing instruments and vendor data formats?
Yes. SpotMap MS is vendor-neutral and works with raw MS files from the major instrument manufacturers via standard exchange formats. The product was built around DDA (data-dependent acquisition) methodology, which is the dominant LC-MS HCP workflow across the industry.
Can we use our own proprietary HCP spectral library, or are we locked into yours?
Either, both, or any combination. You can import your own proprietary FASTA files and spectral libraries; you can use commercially or publicly available libraries; or you can use TotalLab’s AI-powered HCP identity library with built-in threat scoring. Most teams end up using a mix.
How long does it take to get to a first result?
For a typical CHO HCP analysis, point SpotMap MS at your FASTA file and your raw MS files and run. The end-to-end analysis is automated. Customers typically describe the experience as “set it and forget it” — the software produces a comprehensive HCP report without requiring active analyst supervision.
How do we get to 21 CFR Part 11 / Annex 11 compliance?
Through AuditSafe, TotalLab’s universal compliance wrapper. AuditSafe sits around SpotMap MS — and any other software in your stack, including third-party tools like Excel, GraphPad and JMP — and provides secure user authentication, granular permissions, full audit logs, electronic signatures and image authenticity checks. It is designed to meet 21 CFR Part 11, EudraLex Annex 11, GAMP and ALCOA+ requirements. For regulated installations, TotalLab provides an in-person IQ/OQ package as part of the software validation pathway.
We use ELISA for HCP today. Do we need to abandon it to use SpotMap MS?
No. ELISA and LC-MS are orthogonal techniques and most regulated HCP programs need both. SpotMap MS sits alongside PlateLogic (for vendor-neutral ELISA analysis) and SpotMap 2D (for HCP antibody coverage by 2D-PAGE / 2D Western). Many teams use the LC-MS results to validate which HCPs their ELISA antibody set is actually picking up — and which it isn’t.
How does it compare on price to Byos, BioPharma Finder or Genedata?
TotalLab quotes directly, in GBP, USD or EUR depending on your purchasing entity. Pricing depends on number of seats, regulated vs research deployment, IQ/OQ inclusion and support level — book a demo and we’ll give you a like-for-like quote against your current platform.
Is there a free trial?
Yes. The free trial gives you the full software and a guided walk-through with a TotalLab application scientist who has run HCP analysis on real biotherapeutic data. You’ll typically have a clear answer on whether it fits your workflow within a single working session.
Who are TotalLab and how long have they been doing this?
TotalLab is a UK-headquartered scientific software company that has been supporting the world’s largest pharmaceutical companies for over a decade. The company was a long-time supporter of the BEBPA Host Cell Protein Conference and built SpotMap MS in direct collaboration with the leading HCP analysts in industry. Sister products SpotMap 2D and PlateLogic are the standards of the industry for their respective techniques.
See LC-MS HCP analysis built for HCP
Book a 30-minute walkthrough with a TotalLab application scientist, or start a free trial today and run the comparison on your own data.
